
Although a technically challenging process, spray drying is a mature, well understood technique capable of transforming solutions or suspensions into solid particles. Although this process has been widely used in diverse industrial fields, it has become more and more demanded in pharmaceutical applications for the production of solid dispersions.
Today’s APIs are increasingly insoluble and that is presenting new problems for formulators looking to manage the bioavailability and dosing of their formulas. As a result, a significant number of therapeutics gaining approval recently possessed poor biopharmaceutical properties that had to be managed through advanced processes and formulation strategies.
Improving the bioavailability of these new and existing drugs is turning out to be big business for contract development and manufacturing organizations (CDMOs) as pharma’s drug developers look to exploit both accelerated new chemical entity (NCE) and existing drug development pathways.
Introduction
Spray drying active pharmaceutical ingredients (APIs) in solution to overcome solubility hurdles requires part craft and great attention to process variables. In this article, Javier Gurrea, a spray drying manufacturing specialist at Ardena, explains how expertly applied spray drying technology offers drug innovators a faster route to higher-performing drugs.
Although a technically challenging process, spray drying is a mature, well understood technique capable of transforming solutions or suspensions into solid particles. Although this process has been widely used in diverse industrial fields, it has become more and more demanded in pharmaceutical applications for the production of solid dispersions (SDDs).
Pharma leveraging SDDs more than ever
Today’s APIs are increasingly insoluble and that is presenting new problems for formulators looking to manage the bioavailability and dosing of their formulas. As a result, a significant number of therapeutics gaining approval recently possessed poor biopharmaceutical properties that had to be managed through advanced processes and formulation strategies.
Improving the bioavailability of these new and existing drugs is turning out to be big business for contract development and manufacturing organizations (CDMOs) as pharma’s drug developers look to exploit both accelerated new chemical entity (NCE) and existing drug development pathways.
Several of the most popular drugs on the market today have had to manage poor solubility and low bioavailability. That trend isn’t slowing either. Pharma industry analysts estimate that as many as 40% of approved drugs and nearly 90% of the developmental pipeline drugs consist of poorly soluble molecules. (1)
For developers, changing formula chemistries and identifying different routes of administration are just a few of the ways the industry is seeking to profit from accelerated drug development routes including 505(b)(2) New Drug Applications (NDAs). The industry is finding that redeveloping existing formulations can quickly improve the therapeutic value of existing drugs to both payer and patient.
Regarding drug bioavailability enhancement, SDSDs have proven to be a highly controllable, flexible manufacturing strategy to improve the solubility of drugs – especially those with low aqueous solubility.
Because each product is unique, a deep knowledge of the key aspects of the formulation and the mechanistic understanding of spray drying process is required.
Understand your evaporation rate inside and out
During spray drying, the heat and mass transfer that takes place determines the characteristics of the particles being formed. This atomization of the solution is a crucial aspect of the process because it generates fine droplets in order to increase the surface area of the liquid exposed to the drying gas (2).
Initial mass transfer is characterized by a constant evaporation rate, equivalent to a pure solvent droplet—because it refers to the evaporation of the solvent on the surface of the droplet. This is followed by the diffusion of the solvent from the core to the particle surface (2). At this moment, the temperature of the particle suddenly increases and the particle formation rate diminishes due to the higher amount of solvent in the drying gas stream. Consequently, the evaporation rate undergoes a sudden decrease due to the droplet viscosity, which can solidify the surface first, hindering the solvent from leaving the interior of the droplet.
It is a key consideration not to be overlooked or dismissed lightly. The evaporation rate is crucial in stabilizing the amorphous form of the drug, as well as the time the particle is in contact with the hot gas may have an impact on the stability of the product obtained. In this sense, although the drying capacity of the gas can be increased by raising the process temperatures, it cannot rise indefinitely to avoid compromising the stability of the solid dispersion.
Tetris and the rush hour effects
It is important to not forget that the drying process takes place in milliseconds and during this brief point in time, different phenomena occur that can and will determine the characteristics of the particle being formed.
First, the droplet mass remains constant until the solvent begins to evaporate. Then, the amount of solvent of the droplet goes down and the solute content concentrates on the surface of the particle. As a result of this ‘concentration gradient’ there is a slight diffusion of solutes towards the nucleus of the particle as well. In this case, if the diffusion rate of solutes is not as fast as the decrease in droplet volume, a ‘crust’ can form on the surface of the particle (3).
As evaporation of the solvent continues, this outer skin can hinder the evaporation of the solvent from the core of the particle and, depending on the resistance and thickness of the crust formed, the particle can inflate or burst resulting in hollow or porous particle due to the internal pressure. This phenomenon is commonly known as ‘rush hour effect’ (4).
On the contrary, if the evaporation rate is low, the solute particles have enough time to migrate to the core of the particle during the solvent removal, resulting in denser and smaller particles (‘tetris effect’) (See Figure 1).
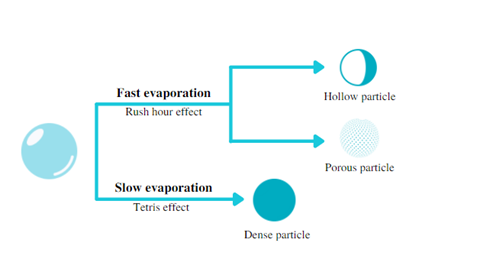
Figure 1. Effect of evaporation rate on transition from droplet to particle.
Fortunately, these phenomena can be explained by a simple equation referring to the Peclet number (Pei) equation which relates the variables that influence the characteristics of the resulting particles (5).

- k is the evaporation rate
- Di is the diffusion coefficient of the solute
For a given solution composition, a low evaporation rate (low Pei) results in smaller and denser particles while a very fast evaporation rate (solvents with low boiling point, high Pei) provides particles of more volume, porous, less dense and with an enrichment of solutes on their surface.
Additionally, the solvent characteristics can also affect the distribution of the particle components, as can be seen in the equation, that includes the diffusion coefficient of the solute in the solvent.
When values of diffusion coefficient are low (corresponding to a higher Pei) there is an enrichment in solutes on the particle surface because the particle components diffuse more slowly than the particle size decrease during the solvent evaporation. In contrast when diffusion coefficients are high (lower Pei) the components will be uniformly distributed along the particle (3,6,7).
All things considered, by manipulating the key process inputs of spray drying, the properties impacting the dissolution behaviour such as morphology and particle size, as well as density and flowability (relevant to process quality and throughput goals in downstream processes like tableting or capsule filling) can be modulated.
Manage spray drying variables for best results
Manufacturers with experience have found that the better technicians are at leveraging a series of critical spray-drying process parameters, the more capable they are of generating desired particle morphologies. Breaking it down, precise particle formation control requires a thorough evaluation of both feed solution variables and process parameters:
Feed solution variables
- Polymer content. The evaporation kinetics is influenced by the polymer concentration in the solution, which will result in a given solution viscosity. On the other hand, the miscibility of both components, and the potential to obtain a homogeneous system, is determined by the API-polymer ratio (8).
- Solids content. Typical solids content used in amorphous solid dispersions are within the range of 10% to 30%. This solids content is inversely proportional to the evaporation rate (9). Regarding desired particle size, low-concentrated solutions generally produce small spherical particles (of high hygroscopicity and concentrated solutions often result in larger particles with a rough surface and high porosity (3,10).
- Solution stability. This variable requires close examination, especially when large commercial volumes of solution are prepared, involving a large period of time between its preparation and its drying process, in order to avoid nucleation and crystalline growth (11).
Process parameters
- Liquid feeding rate. This parameter dictates the time in which the particles are in the drying chamber, as well as the amount of solvent present in the gas stream and the subsequent outlet temperature observed. It is also directly proportional to the particle size and some authors have described that this feed rate could be inversely proportional to the solubility enhancement of the active ingredient (12).
- Inlet temperature. Because inlet temperature has been postulated to be directly proportional to the obtained glass-transition temperature (Tg), it is inversely proportional to the crystallinity of the drug (13). High inlet temperatures can generate larger particles and may cause solvent entrapment in its core, resulting in the subsequent destruction of its outer skin while lower inlet temperatures, generate smaller denser particles with a rough surface (3,14).
- Outlet temperature. Two aspects of outlet temperature should never be overlooked. If the outlet temperature is above the Tg of the product, it can adhere to the walls of the equipment due to the sticky characteristics of the compound, reducing process yield. Similarly, an outlet temperature too low will cause a high level of residual solvent in the product, compromising its stability (15).
- Type of gas and flow rate. On the one hand, the type of gas used can influence the particle size. Gases with low density, such as nitrogen, result in smaller particle size (2,16). On the other hand, the higher the gas flow rate, the smaller the particle size obtained during the process and, in addition, it has been observed that working in open cycle produces higher yields than working in closed cycle (2,17).
- Type of atomizers used. Depending on the design of the atomizing system, particles with different properties can be obtained using:
Rotary/centrifugal atomizers: These devices use a rotating disk to break the liquid stream into small droplets that are projected towards the walls of the drying chamber thanks to centrifugal force (18).
Bi-fluid nozzles: This is the most common type of atomizer employed in the pharmaceutical field. In these devices the liquid is put in contact with a gas stream resulting in a disintegration of the liquid into fine droplets. The characteristics of the atomization will be influenced by the characteristics of the solution or suspension and the gas used (density, viscosity, pressure, etc.) (19).
Pressure nozzles: This type employs hydraulic pressure to break the liquid stream through a nozzle, where a series of spiral-shaped inserts break the solution into small droplets. One advantage of these atomizers is that they allow the obtention of larger particles that facilitates the subsequent downstream process without needing to perform a dry granulation step to achieve the optimum flow and density features (20).
Programs run better with systematic Quality by Design
Although spray drying can be a challenging technology to master it has fast become the preferred way for drug developers to overcome the limitations of APIs with poor aqueous solubility due to its applicability to obtain amorphous solid dispersions or to dry nanosuspensions, for instance. In support of quality and reliability in process the industry is increasingly introducing spray drying in a more systematic and empirical way following ICH Q8 (21) guidelines and its primary Quality by Design (QbD) approach.
Having a deep knowledge of all the parameters that influence the process and their potential impact on particle formation is the initial and key step in starting a successful drug manufacturing program for tricky, insoluble formulations. The process is sophisticated and program planning requires a precision approach. Ardena has a long experience leveraging spray drying to meet highly potent drug products therapeutic and manufacturing goals. As a CDMO we offer access to all current GMP spray drying capabilities as well as complete manufacturing services in support of all clinical and commercial phase drug development.
References
- Kalepu S, Nekkanti V. Insoluble drug delivery strategies: review of recent advances and business prospects. Acta Pharmaceutica Sinica B. 2015; 5(5):442-53.
- Singh A, Van den Mooter G. Spray drying formulation of amorphous solid dispersions. Adv Drug Deliv Rev. 2016; 100:27–50.
- Paudel A, Worku ZA, Meeus J, Guns S, Van den Mooter G. Manufacturing of solid dispersions of poorly water soluble drugs by spray drying: Formulation and process considerations. Int J Pharm. 2013 Aug 30;453(1):253–84.
- Pai DA; Vangala VR;, Ng JW;, Tan RBH. Resistant maltodextrin as a shell material for encapsulation of naringin: Production and physicochemical characterization Item Type Article. 2015.
- Vehring R, Foss WR, Lechuga-Ballesteros D. Particle formation in spray drying. J Aerosol Sci. 2007 Jul;38(7):728–46.
- Osman A, Goehring L, Patti A, Stitt H, Shokri N. Fundamental Investigation of the Drying of Solid Suspensions. Ind Eng Chem Res. 2017 Sep 20;56(37):10506–13.
- Lintingre E, Lequeux F, Talini L, Tsapis N. Control of particle morphology in the spray drying of colloidal suspensions. R Soc Chem. 2016;12(36):7435–44.
- Wang S, Langrish T. A review of process simulations and the use of additives in spray drying. Food Res Int. 2009 Jan;42(1):13–25.
- Miller, D.A., Gill, M. Spray-drying technology. Formula Poorly Water Soluble Drugs SpringerNew York. 2012; 3:363–442.
- Littringer EM, Mescher A, Eckhard S, Schröttner H, Langes C FM. Spray Drying of Mannitol as a Drug Carrier—The Impact of Process Parameters on Product Properties. Dry Technol. 2012 Jan;30(1):114–24.
- Lindfors L, Forssén S, Westergren J, Olsson U. Nucleation and crystal growth in supersaturated solutions of a model drug. J Colloid Interface Sci. 2008 Sep 15;325(2):404–13.
- Sahoo NG, Abbas A, Judeh Z, Li CM, Yuen K-H. Solubility Enhancement of a Poorly Water-Soluble Anti-Malarial Drug: Experimental Design and Use of a Modified Multifluid Nozzle Pilot Spray Drier. J Pharm Sci. 2009 Jan 1;98(1):281–96.
- Albers J, Matthée K, Knop K, Kleinebudde P. Evaluation of predictive models for stable solid solution formation. J Pharm Sci. 2011.
- Dobry DE, Settell DM, Baumann JM, Ray RJ, Graham LJ, Beyerinck RA. A model-based methodology for spray-drying process development. J Pharm Innov. 2009.
- Thybo P, Hovgaard L, Lindeløv JS, Brask A, Andersen SK. Scaling up the spray drying process from pilot to production scale using an atomized droplet size criterion. Pharm Res. 2008 Jul 11;25(7):1610–20.
- Özbilen S. Influence of atomising gas on particle characteristics of Al, Al–1 wt-%Li, Mg, and Sn powders. Powder Metall. 2000 Feb 19;43(2):173–80.
- Wang A, Lu Y, Zhu R, Li S, Ma X. Effect of process parameters on the performance of spray dried hydroxyapatite microspheres. Powder Technol. 2009 Apr 4;191(1–2):1–6.
- Huang LX, Kumar K, Mujumdar AS. A comparative study of a spray dryer with rotary disc atomizer and pressure nozzle using computational fluid dynamic simulations. Chem Eng Process Process Intensif. 2006 Jun 1;45(6):461–70.
- Masters K (Keith). Spray drying in practice. Charlottenlund: SprayDryConsult; 2002. 464 p.
- Mujumdar AS. Handbook of industrial drying. CRC/Taylor & Francis; 2007.
- International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use. Pharmaceutical Development Q8(R2).
